抗体偶联药物(ADC)纯化选择
抗体药物偶联物(ADC)是通过偶联臂将抗体和小分子药物相结合,可杀死或抑制癌细胞等特定细胞的抗体药物。到2021年10月为止,除了美国FDA已经批准上市的12个品种以外,还有3种具有类似结构的免疫毒素偶联物(重组)也获得了批准。随着研发的不断推进,目前还有约150种ADC药物正处于临床试验阶段。
在ADC的纯化工艺中常用到的分离方法,一种是在抗体上结合药物之后,通过疏水层析(HIC)分离出结合小分子药物数量不同的ADC;另一种是通过尺寸排阻层析(SEC)根据分子尺寸的差异,分离ADC的多聚体和未反应试剂。
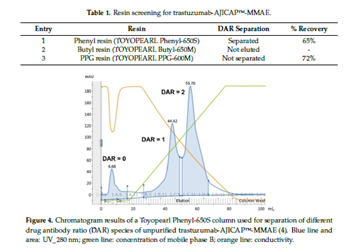
利用疏水填料进行纯化示例
HIC column (0.8 *10 cm, 5 mL) ToyoPearl
Phenyl-650S HIC resin (Tosoh Bioscience)
buffer A :50 mM sodium phosphate pH 7.0, 2 M
sodium chloride (NaCl)
buffer B :50 mM sodium phosphate pH 7.0, 20%
isopropyl alcohol (IPA) v/v).
loading:25mg/ml sample 25ml+25ml buffer A
Linear gradient: buffer A to buffer B
如需了解多详情请参考一下文献:
1.
Y. Matsuda et al., A purification strategy utilizing
hydrophobic interaction chromatography to obtain homogeneous species from a
site-specific antibody drug conjugate produced by AJICAP™first generation
antibodies, 2020, 9, 16,
2.
S. J. Walsh et al., Site-selective modification
strategies in antibody-drug conjugates, Chem. Soc. Rev., 2021, 50, 1305
3.
Q. Dean et al., Targeting cancer with antibody-drug
conjugates; Promises and challenges, MAbs. 2021; 13(1): 1951427,
4.
J. T. W. Tong et al., An insight into FDA approved
antibody-drug conjugates for cancer therapy, Molecules, 2021, 26, 5847,
| 
